Neurotrophic Keratitis
When Dry Eye treatments fail, could Neurotrophic Keratitis (NK) be the reason?
Some of the most challenging ocular surface cases can have overlapping causes. Since early-stage NK can look like Dry Eye,1 identifying it as a possibility may help bring the full picture into focus.
Understanding NK and the Power of Early Intervention:
NK is a corneal degenerative disease characterized by a reduction of corneal sensitivity.2 In NK, corneal innervation by the trigeminal nerve is impaired.2
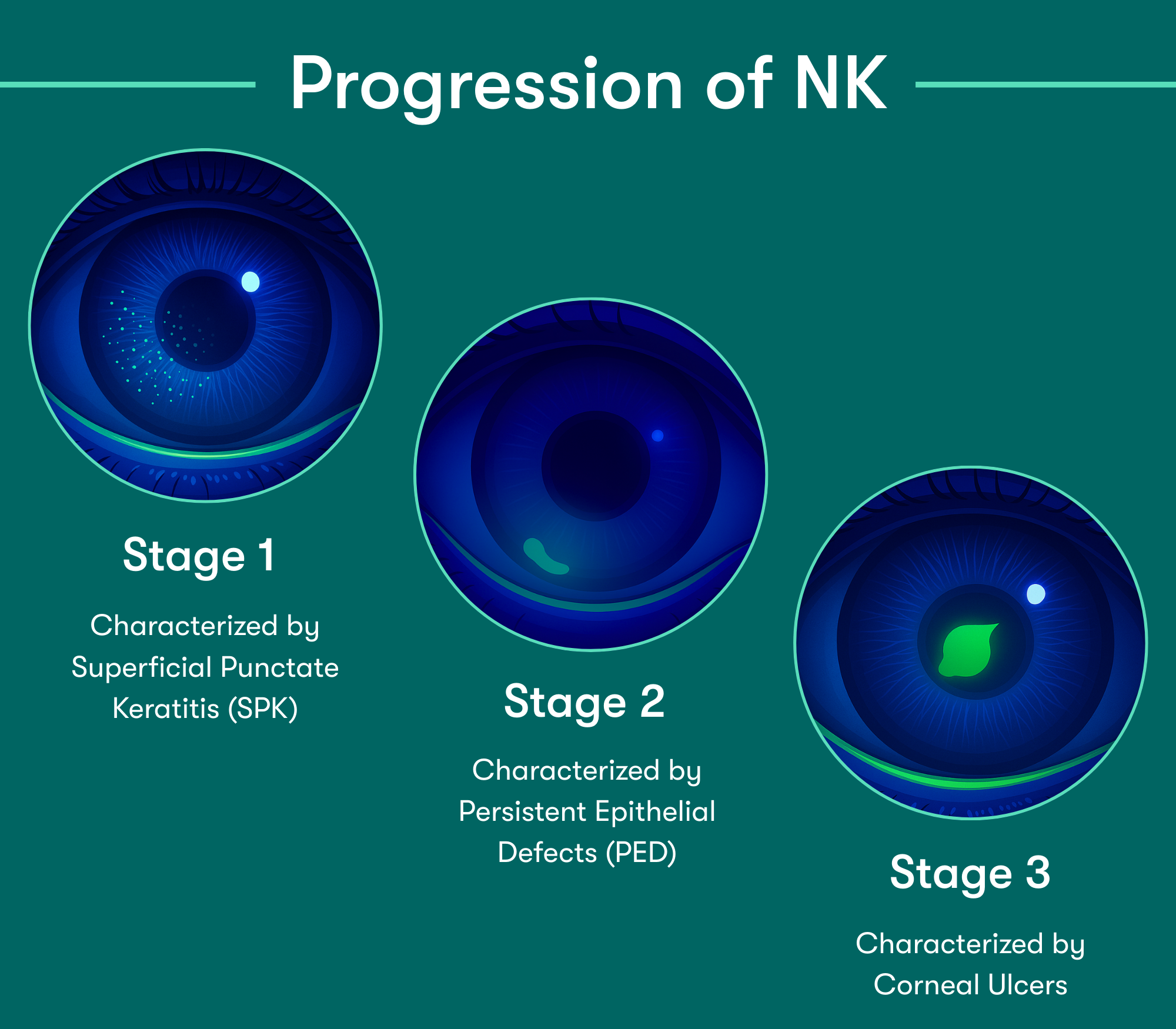
According to the Mackie classification, Stage 1 NK begins as early as Punctate Epithelial Keratitis (PEK).3 Lack of early detection can lead to corneal epithelial breakdown, impairment of healing, and development of corneal ulceration, melting, and perforation.4
Early detection & treatment are key to preventing further epithelial damage. Corneal sensitivity testing can help identify patients who may have underlying nerve damage and don’t respond to standard dry eye treatments.3 Detecting reduced sensitivity early can point to early stage Neurotrophic Keratitis and guide more targeted therapy to support healing and protect the cornea. Reduced corneal sensation affects nearly 10% of all eyes, and 30% of eyes with signs & symptoms of DED.1
How can I test for NK?
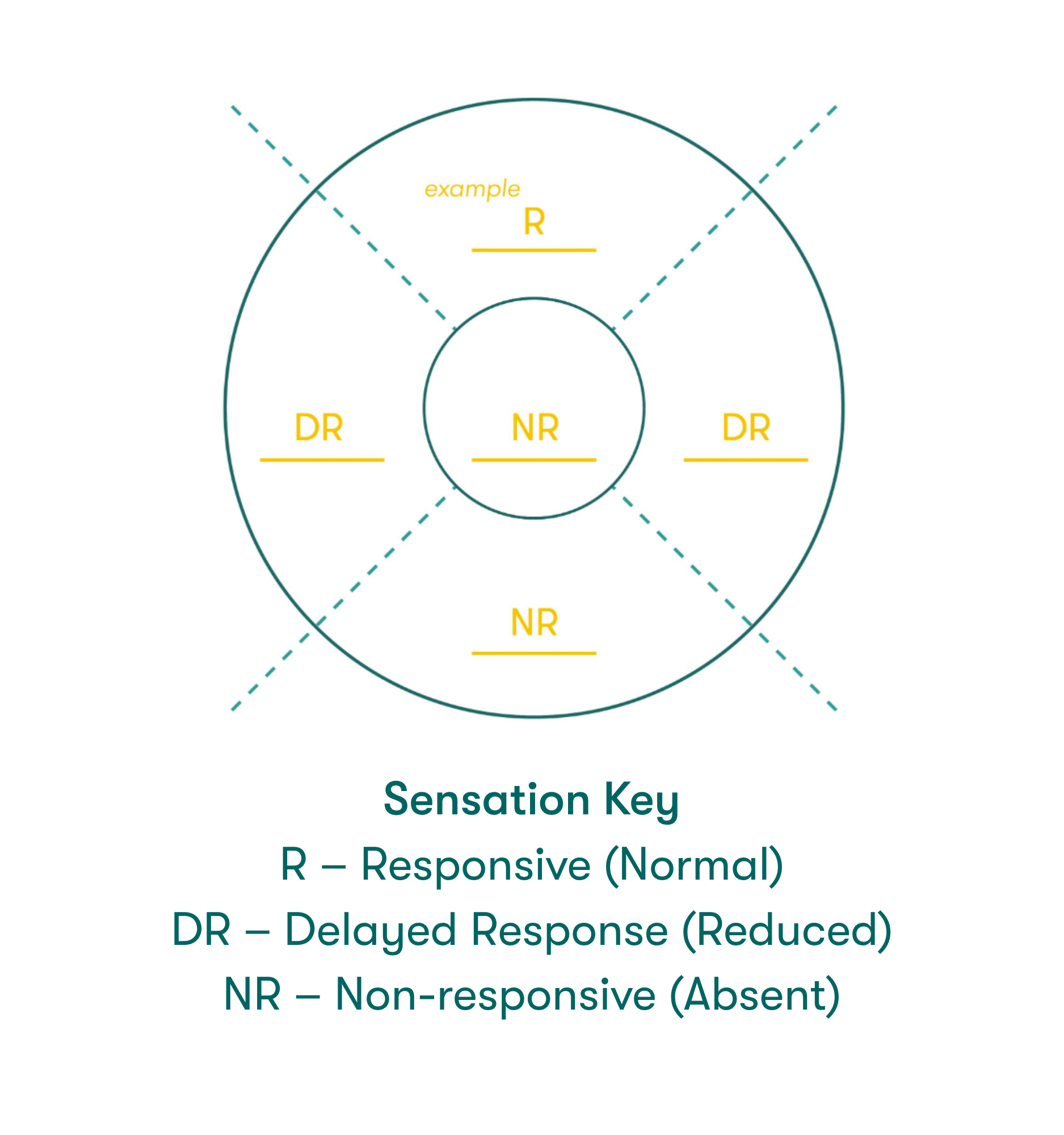
Corneal sensitivity testing can help confirm the diagnosis of NK & assess the severity of corneal nerve impairment. Corneal sensitivity can be measured in two ways:1
- Qualitatively, by touching the central & peripheral cornea with a cotton thread or un-waxed & unflavored dental floss
- When the thread or floss gently touches the cornea, patients with a normal cornea show a blink reaction and can describe the sensation of touch, while patients with loss of corneal sensitivity do not react
- Quantitatively, using a corneal aesthesiometer
The Sensation Key graphic on the right can be labeled with level of sensation to help with disease diagnosis.

Limited treatment options for NK? We have two!
Why Cryopreserved Amniotic Membrane for NK?
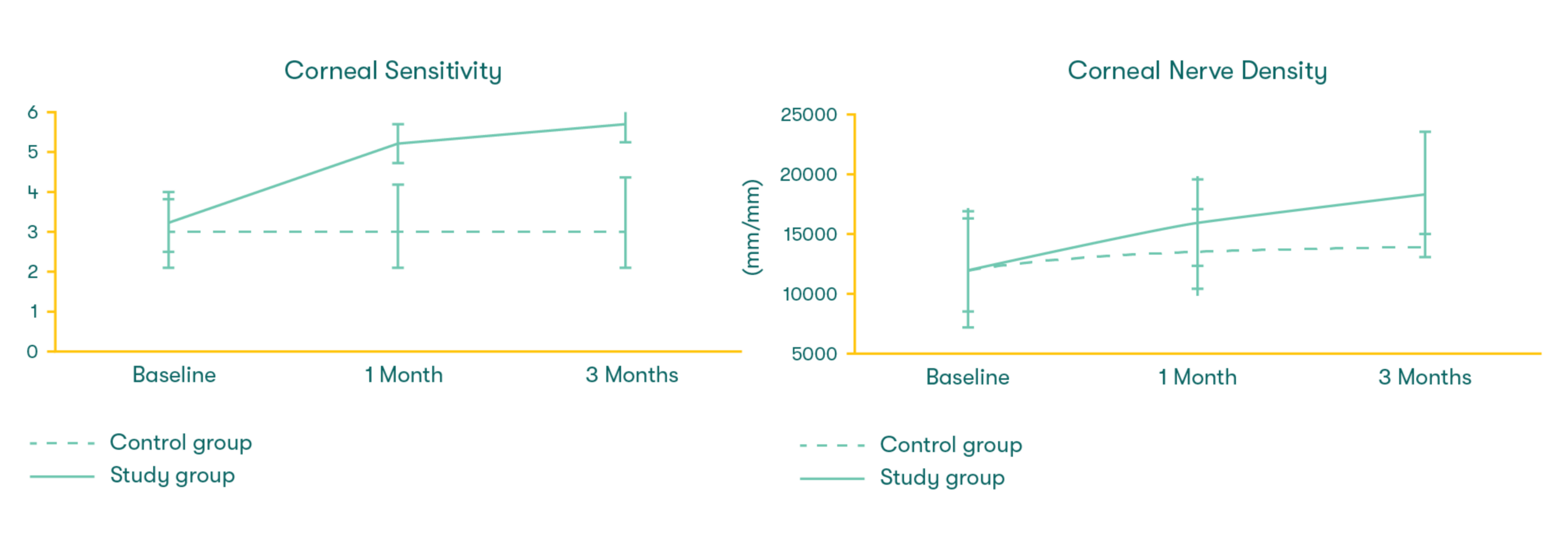
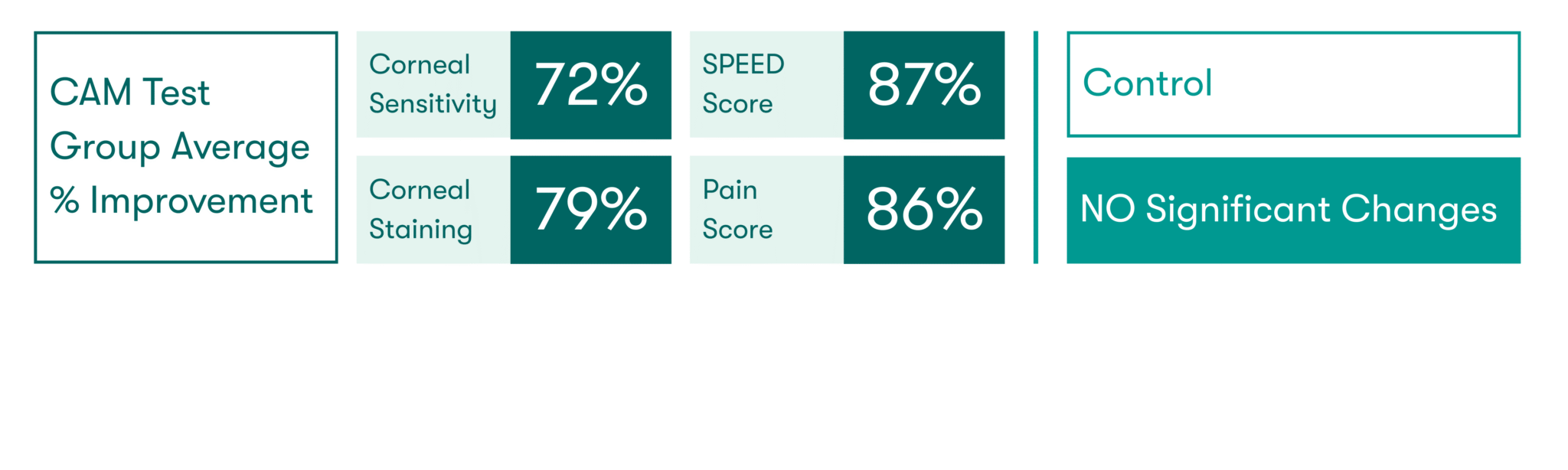
As featured in Thomas John’s clinical study, there was a significant increase in corneal sensitivity from 1 to 3 months. This improvement was significantly correlated with the increase of corneal nerve density.7
Interested in the data?
Read the full Thomas John et al., 2017 study on Cryopreserved Amniotic Membrane and ocular surface health.
The American Academy of Ophthalmology (AAO) recognizes Prokera, a self-retained Cryopreserved Amniotic Membrane (CAM) device, as a Stage 1 treatment option for Neurotrophic Keratitis (NK).2
CAM preserves innate complexes found in amniotic membrane tissue that help support the regeneration of corneal surface.8,9 In a clinical study, CAM is shown to reduce signs and symptoms in ocular surface disease and restore corneal nerve density, helping improve corneal sensitivity in patients with Dry Eye Disease. 7
Products like Prokera and CAM360 AmnioGraft give physicians a convenient, in-office treatment for patients with NK. Early intervention using Cryopreserved Amniotic Membranes may protect the cornea and support nerve regeneration.
Do you want to learn more about CAM360 AG & Prokera?